We use cookies to give you the best online experience. By using our website you agree to our use of cookies in accordance with our Cookie Policy
AccuPower® TB & MDR Real-Time PCR Kit
AccuPower® TB&MDR Real-Time PCR Kit is an in vitro diagnostic kit designed for the simultaneous detection of Mycobacterium tuberculosis (MTB) and muti-drug resistant (MDR) TB DNA in human samples such as sputum, bronchoalveolar lavage (BAL), and culture specimen through Real-Time PCR.
Multidrug-resistant TB (MDR-TB) is caused by an organism that is resistant to at least isoniazid (INH) and rifampicin (RIF), the two most potent TB drugs. These drugs are used to treat all persons with TB disease. Spontaneous mutations in the M. tuberculosis genome can give rise to proteins that make the bacterium drug resistant, depending on the drug action. Almost 2~3% of new TB patients have MDR-TB. For the effective treatment of TB and MDR-TB, early detection is the top priority.
Features and Benefits
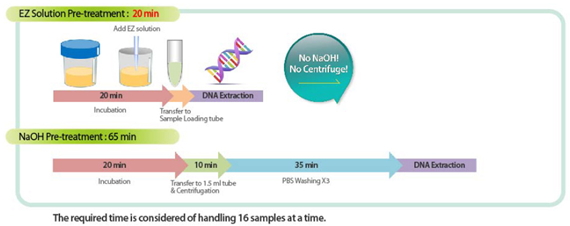
- Signature Convenience: Multiplex detection of TB and MDR-TB (13 mutant sites, 36 mutations). Simplified procedure of sputum pretreatment with EZ Solution.
- Enhanced Sensitivity & Specificity: Bioneer’s proprietary HotStart technology accomplishes high sensitivity and specificity.
- Remarkable Stability: Vacuum-dried premix allows stable and reproducible results.
- High Quality: All diagnostic kits manufactured by Bioneer are under strict quality control guidelines.
- Sputum Pre-treatment: EZ Solution vs NaOH
Specifications
| Specimen Type | Sputum, bronchoalveolar lavage (BAL), culture specimen |
| Kit Contents | PCR Premix (RIF/TB&INH), PC, IPC, NTC, DEPC D.W. |
| Instrumentation | ExiStation™ ™ Universal Molecular Diagnostic System or Exicycler™ 96 |
| Tests | 48 |
Performances
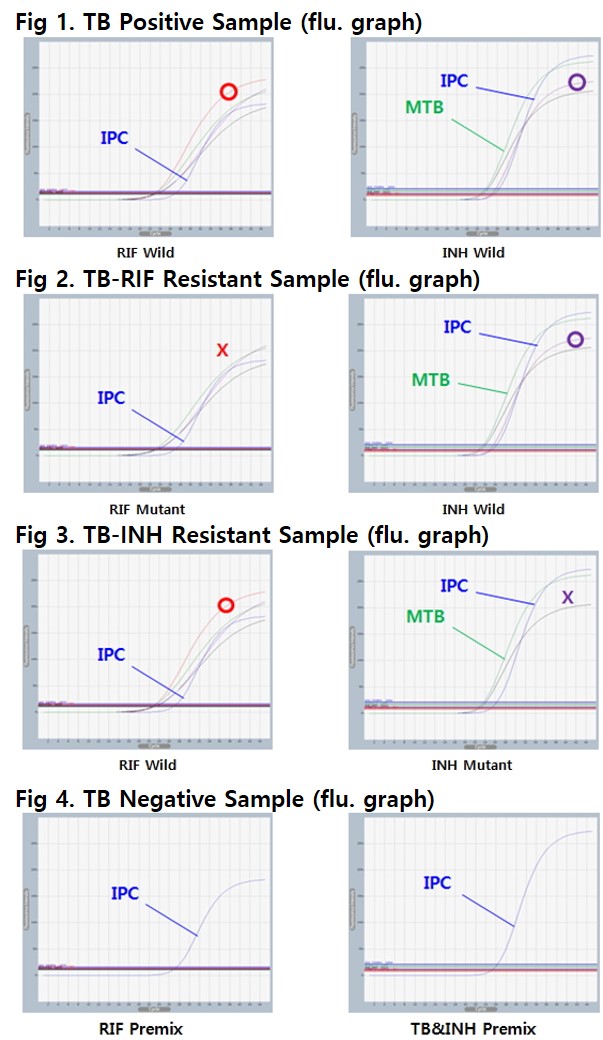
AccuPower® TB&MDR Real-Time PCR Kit includes Positive Control (PC), No Template Control (NTC) and Internal Positive Control (IPC) for accurate and reliable diagnosis of MTB and MDR-TB. (Wild type : All detection of TB and MDR-TB, Mutant type : No detection of more than 1 MDR-TB)
Ordering Information
| Cat. No. | Product Description |
| TBMDR-1111 | AccuPower® TB&MDR Real-Time PCR Kit (48 tests) |
Related Products
| Cat. No. | MDx Diagnostic Kit |
| MTB-1111 | AccuPower® MTB Real-Time PCR Kit (96 tests) |
| MTN-1111 | AccuPower® MTB&NTM Real-Time PCR Kit (96 tests) |
| NTM-1111 | AccuPower® NTM Real-Time PCR Kit (96 tests) |
| Cat. No. | Instrument |
| A-2060 | Exicycler™ 96 Real-Time Quantitative Thermal Block |
| A-2200 | ExiStation™ Universal Molecular Diagnostic System |
| A-5050 | ExiPrep™16 Dx Automated Nucleic Acid Extraction System (16 tests) |
| Cat. No. | Nucleic Acid Extraction Kit |
| K-4418 | ExiPrep™ Dx Mycobacteria Genomic DNA Kit (96 tests) |
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
Contact Us
E-mail : sales@bioneer.com