We use cookies to give you the best online experience. By using our website you agree to our use of cookies in accordance with our Cookie Policy
AccuPower® RV Kit (ExiStation™ FA 96)
AccuPower® RV Kit(ExiStation™ FA 96) is an in vitro diagnostic kit designed for the qualitative detection and differentiation of SARS-CoV-2, Influenza A, Influenza B, and RSV A/B.
AccuPower® RV Kit (ExiStation™ FA 96) is a multiplex Real-Time PCR solution designed for the simultaneous or single qualitative detection and differentiation of SARS-CoV-2, Influenza A, Influenza B, and RSV A/B. Used with the Automated ExiStation™ FA 96, it streamlines the workflow from nucleic acid extraction to amplification for enhanced laboratory efficiency. As these respiratory viruses can present with similar clinical symptoms, accurate differential detection is essential for timely diagnosis and appropriate patient management. The assay delivers reliable molecular testing performance using human nasopharyngeal swab specimens. It is intended for use by healthcare professionals in clinical laboratory environments.
Features and Benefits
- Broad detection coverage
Detects over 40 Omicron subvariants and major influenza subtypes and lineages, including Influenza A H1N1, H3N2, Influenza B Yamagata, and Influenza B Victoria, with high sensitivity. - Enhanced Convenience & Stability
Ready-to-use premixed kit with extended shelf life, enabling room temperature shipping - High sensitivity and specificity
Dual Hot Start technology and UDG support reliable assay performance. - Compatible with ExiStation™ FA 96
Designed for use with the fully automated sample-to-result system to reduce hands-on time and minimize user error.
Specifications
| Specimen Type | Nasopharyngeal swab |
| Kit Contents | RVP Premix, RVP PC, RVP NTC, Sealing Film, Quick Manual |
| Instrumentation | ExiStation™ FA 96 (A-2500-1) |
| Tests/Kit | 96 |
Performances
AccuPower® RV Kit (ExiStation™ FA 96) test results for clinical samples.
An internal Positive control is used to validate the PCR results of each specific sample.
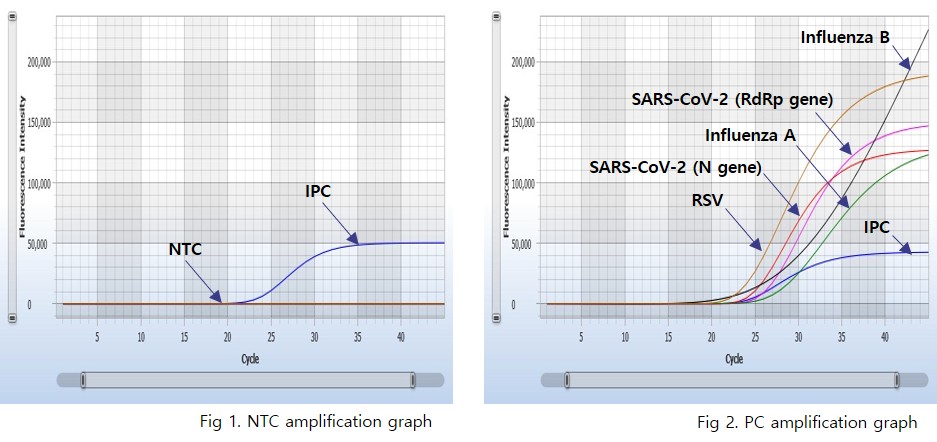
AccuPower® RV Kit (ExiStation™ FA 96) includes negative control and positive control to verify the validity of the experiment.
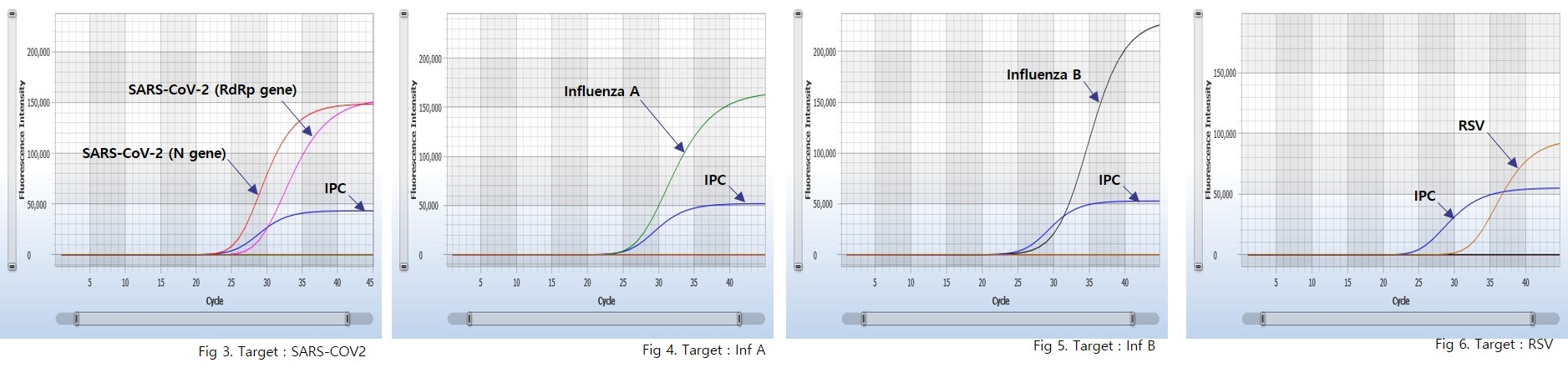
User centric ExiStation™ FA 96 software automatically analyzes the test results based upon the Ct (threshold cycle) value.
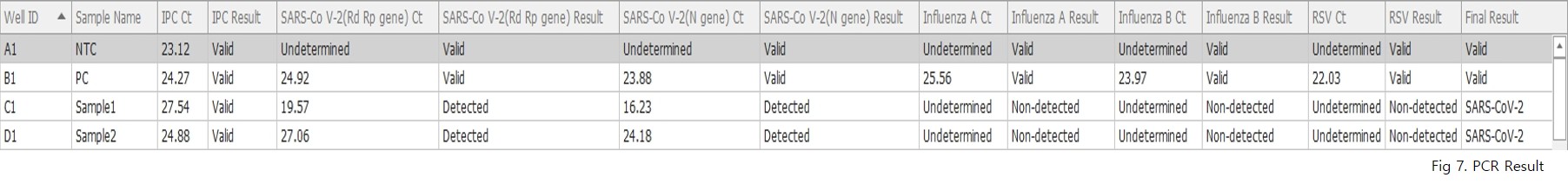
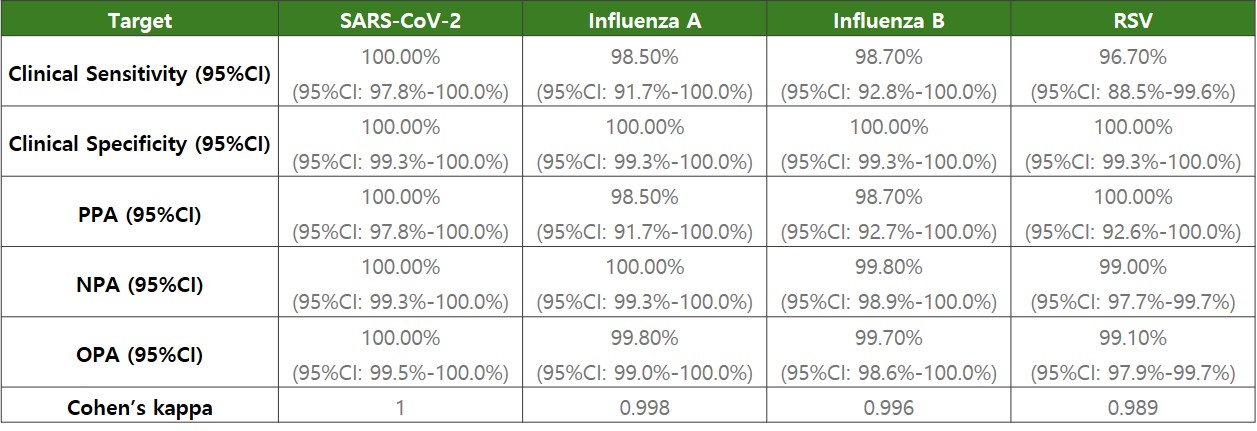
Clinical evaluation results of AccuPower® RV Kit (ExiStation™ FA 96).
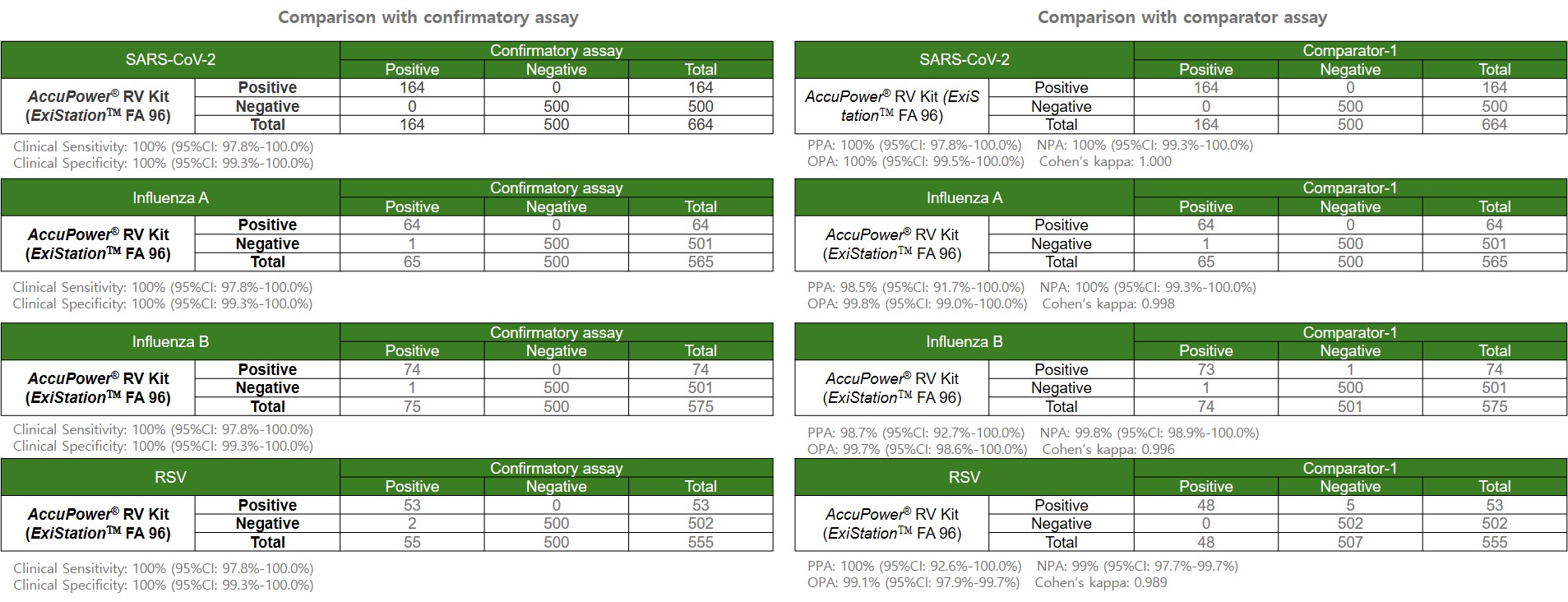
The evaluation was conducted using 859 residual clinical specimens from Europe.
Clinical Evaluation Results
Ordering Information
| Cat. No. | Product Description | Certification |
| RVP-3116 | AccuPower® RV Kit (ExiStation™ FA 96) | CE-IVDR Class B |
Related Products
| Cat. No. | Instrument |
| A-2500-1 | ExiStation™ FA 96 |
| Cat. No. | Nucleic Acid Extraction Kit |
| K-4815 | ExiStation™ FA DNA/RNA Kit(V3) |
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
Contact Us
E-mail : sales@bioneer.com
v0.0 / 2019-03-05
