We use cookies to give you the best online experience. By using our website you agree to our use of cookies in accordance with our Cookie Policy
IRON-qPCR™ RFIA Kit

IRON-qPCR™ RFIA Kit (RFIA-1113) is an in vitro diagnostic assay for the qualitative detection of Mycobacterium tuberculosis (MTB), enabling the simultaneous identification of the MTB complex and MDR-/Pre-XDR-TB resistance profiles in a single test.
The IRON-qPCR™ RFIA Kit is a CE-IVD marked in vitro diagnostic assay designed for the rapid and comprehensive detection of Mycobacterium tuberculosis (MTB) and its drug resistance profiles. The kit provides highly sensitive and specific detection across four major classes of anti-TB drugs: Rifampicin, Isoniazid, Fluoroquinolones, and Aminoglycosides
Utilizing advanced real-time PCR technology, this All-in-One test simultaneously identifies wild-type MTB and screens for 56 key mutations associated with Multi drug-Resistant (MDR) and pre-Extensively Drug-Resistant (pre-XDR) TB directly from pre-treated sputum or culture specimens. Accurately and quickly diagnosing drug-resistant TB is essential for establishing timely, targeted treatment strategies and improving patient outcomes.
The kit is intended for use on the dedicated IRON-qPCR™ system. The standard workflow features a highly streamlined, user-friendly process with convenient cartridge loading, delivering accurate and actionable results in just 40 minutes—offering fast diagnostic pathway for comprehensive TB patient care.
Features and Benefits
-
Comprehensive All-in-One Test: Detects wild-type MTB and screens 56 drug-resistance mutations (MDR-TB & pre-XDR-TB) across 4 major drug classes in a single assay.
-
Ultra-Rapid 40-Min Workflow: Delivers highly accurate results in just 40 minutes, providing a significantly faster diagnostic pathway than conventional methods.
-
Advanced Mutation Detection: Prevents false-negative RIF resistance results by successfully identifying critical mutations, including the elusive rpoB I491F
-
Proven Diagnostic Excellence: Achieved a 100% concordance rate in the QCMD EQA programme, with over 99% sensitivity and specificity.
-
Enhanced Stability & Convenience: Features an extended 24 months shelf life and allows for room temperature storage (15~35°C), completely eliminating cold-chain logistics.
Specifications
| Specimen Type | Sputum, Culture specimen |
| Kit Contents | All-in-One Cartridge (DNA Extraction & PCR Reaction Reagent) |
| Instrumentation | IRON-qPCR™ |
| Tests/kit | 20 |
Performances
- Material: MDR-TB, pre-XDR-TB Sample panel from Foundation for Innovative New Diagnostics (FIND)
- Test site: ITRC(International Tuberculosis Research Center)
- Comparison method: Sequencing
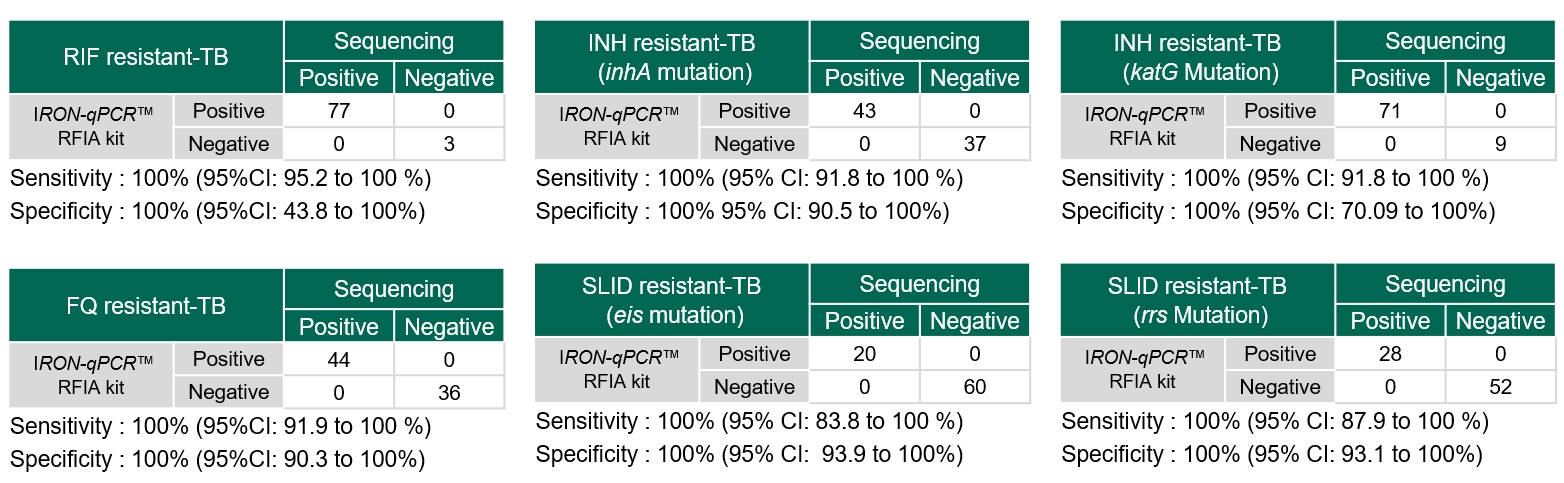
The test result confirmed IRON-qPCR™ RFIA Kit’s ability to determine the presence of each TB treatment drug resistant genes(associated with MDR-TB and pre-XDR-TB) with high sensitivity and specificity.

Achieved 100% concordance in the QCMD EQA programme, demonstrating reliable detection of TB and multi-drug resistance.
Ordering Information
| Cat. No. | Product Description | Certification |
| RFIA-1113 | IRON-qPCR™ RFIA Kit | CE-IVDD |
Related Products
| Cat. No. | Instrument |
| A-2600 | IRON-qPCR™ |
| Cat. No. | Pre-treatment |
| K-4174 | E-z solution II |
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
Contact Us
E-mail : sales@bioneer.com
v0.0 / 2019-03-
v0.0 / 2019-03-05