We use cookies to give you the best online experience. By using our website you agree to our use of cookies in accordance with our Cookie Policy
IRON-qPCR™ RV Kit
IRON-qPCR™ RV Kit (QRV-1113) is an in vitro diagnostic reagent for the qualitative detection of respiratory viruses, including SARS-CoV-2, Influenza A/B, and RSV-A/B, in nasopharyngeal swab specimens using the IRON-qPCR™ System.
The IRON-qPCR™ RV Kit (QRV-1113) is an in vitro diagnostic reagent designed for use with the IRON-qPCR™ System (A-2600) for the qualitative detection of SARS-CoV-2, Influenza A, Influenza B, RSV A, and RSV B in human nasopharyngeal swab specimens. The kit provides an automated workflow from nucleic acid extraction to multiplex Real-Time PCR and result interpretation after sample loading.
Clinical specimen evaluation has confirmed high diagnostic sensitivity and specificity for each target. Additionally, the main kit can be stored at 15–35°C, while the QRV PC/NC control set is stored separately at 2–8°C.
Features and Benefits
- Simultaneous detection in a streamlined workflow
Qualitatively detects SARS-CoV-2, Influenza A, Influenza B, RSV A, and RSV B in one streamlined testing process. - Rapid automated workflow
The IRON-qPCR™ system automates key testing steps from nucleic acid extraction and PCR setup to amplification and result interpretation, supporting timely and efficient testing. - Results in around 40 minutes
After cartridge preparation and sample loading, the test runs automatically, helping reduce hands-on time and simplify laboratory workflow. - Validated for human nasopharyngeal swab specimens
Performance has been verified with human nasopharyngeal swab specimens, supporting reliable respiratory virus testing. - Convenient storage and shipment
The kit supports room temperature handling, storage, and distribution for improved operational convenience.
Specifications
| Specimen Type | Nasopharyngeal swab |
| Kit Contents | Test Cartridge(20 tests/kit), User Guide |
| Instrumentation | IRON-qPCR™ (A-2600) |
| Controls | IRON-qPCR™ RV Kit Control Set (QRV-1113C), provided separately |
| Tests/kit | 20 |
Performances
Negative control and positive control are used to verify assay validity according to laboratory quality control procedures.
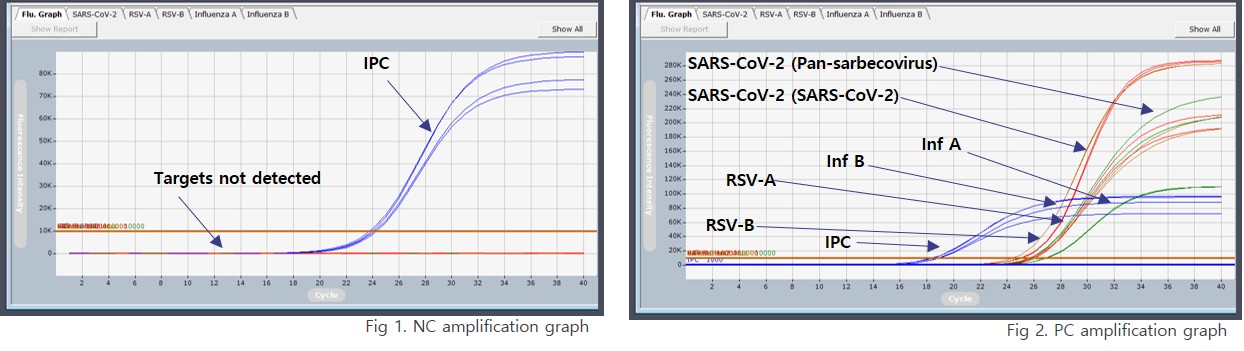
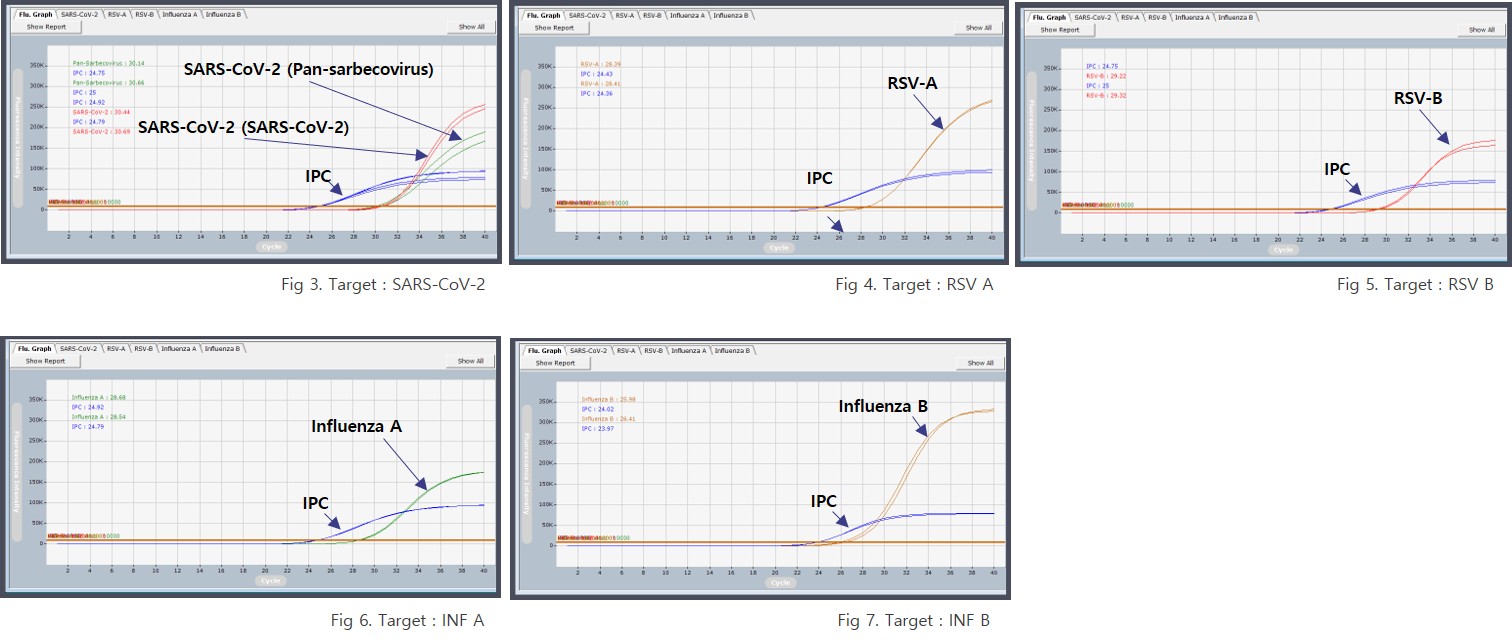
Representative test results of IRON-qPCR™ RV Kit. The assay includes internal process control signals to support result validity assessment for each sample.
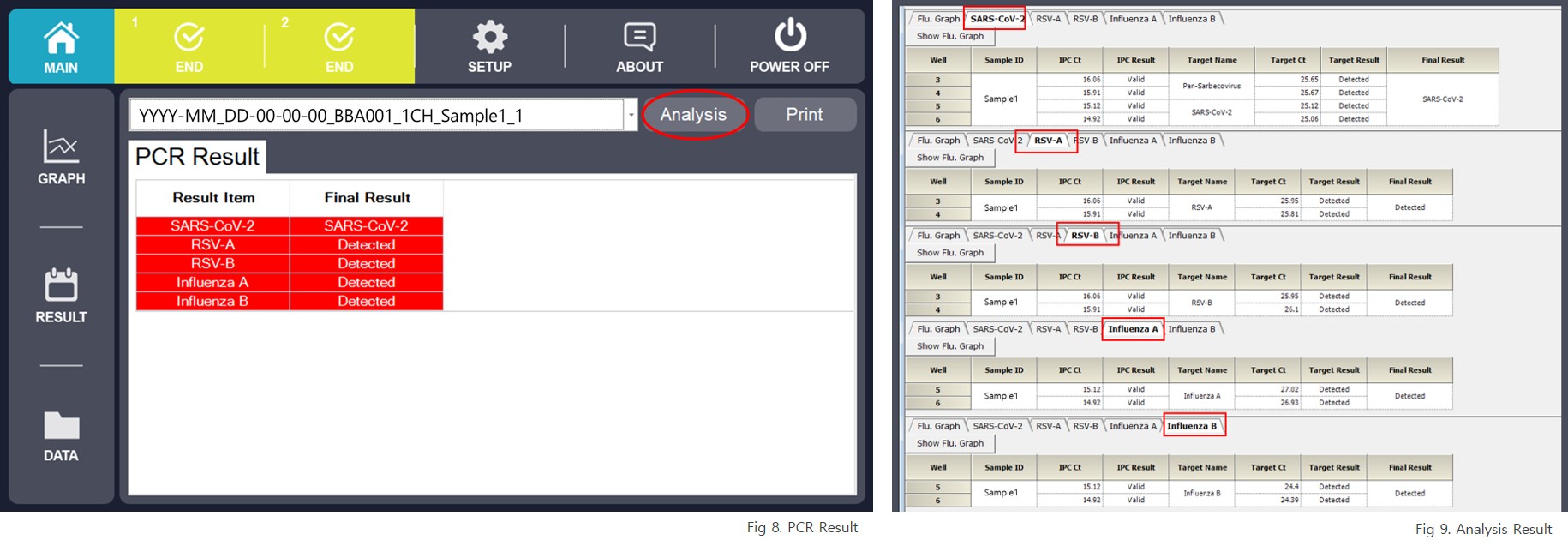
The IRON-qPCR™ software automatically analyzes amplification data and reports qualitative results based on predefined interpretation criteria.
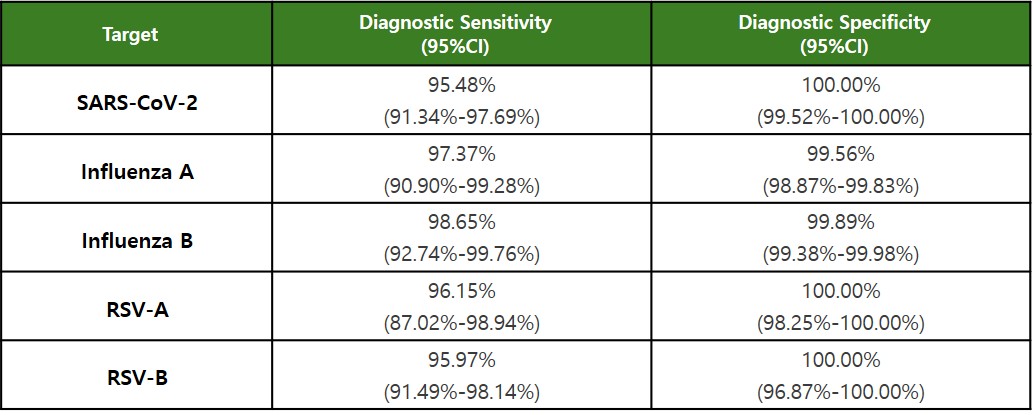
Clinical performance of the IRON-qPCR™ RV Kit was evaluated using residual nasopharyngeal swab specimens collected at a clinical site in France.
Diagnostic sensitivity and specificity were determined by comparison with CE-IVD marked comparator assays.
Clinical Evaluation Results
Ordering Information
| Cat. No. | Product Description | Certification |
| QRV-1113 | IRON-qPCR™ RV Kit | CE-IVDR Class B |
Related Products
| Cat. No. | Instrument |
| A-2600 | IRON-qPCR™ |
| A-2600-A6 | Puncher |
| Cat. No. | Consumables |
| QRV-1113C | IRON-qPCR™ RV Kit Control Set |
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
Contact Us
E-mail : sales@bioneer.com
v0.0 / 2019-03-05
