We use cookies to give you the best online experience. By using our website you agree to our use of cookies in accordance with our Cookie Policy
AccuPower® HIV-1 Quant Kit (ExiStation™ FA 96)
AccuPower® HIV-1 Quant Kit (ExiStation™ FA 96) is an in vitro diagnostic kit designed for the quantification of HIV-1 RNA in EDTA-plasma samples using Real-Time PCR.
AccuPower® HIV-1 Quant Kit (ExiStation™ FA 96) is an in vitro diagnostic kit designed for the quantification of HIV-1 RNA in EDTA-plasma samples using Real-Time PCR, and is fully compatible with ExiStation™ FA 96.
When used with ExiStation™ FA 96, the system enables the processing of up to 96 samples within 120 minutes, supporting high-throughput testing.
The kit provides sensitive and reliable quantification of HIV-1 RNA, developed based on Bioneer's technology and experience from WHO Prequalification HIV-1 Kit :
- Cat. No. HIV-3116 (for ExiStation™ FA 96): Asia's first CE-IVDR Class D
Features and Benefits
- Reliable Viral Load Monitoring: Enables quantification of HIV-1 RNA from 50 IU/mL to 10⁸ IU/mL
- True Full Automation: Compatible with ExiStation™ FA 96, delivering a streamlined workflow from de-capping & re-capping through extraction to PCR
- Fast Turnaround Time: 96 results/hour after initial 120 mins run
- Comprehensive HIV-1 Subtypes Coverage: HIV-1 subtype Group M (A-H, and several CRFs), Group N and Group O are detectable with high sensitivity
- High Sensitivity & Specificity: Bioneer’s proprietary Dual-HotStart™ technology with UDG ensures high detection performance
- Enhanced Convenience & Stability: Ready-to-use premixed kit with 24 months shelf life, enabling room temperature shipping
Specifications
| Specimen Type | EDTA-plasma |
| Kit Contents | PCR Premix, SPC, LPC, HPC, NTC, Sealing film |
| Instrumentation | ExiStation™ FA 96 |
| Tests/kit | 96 |
Performances
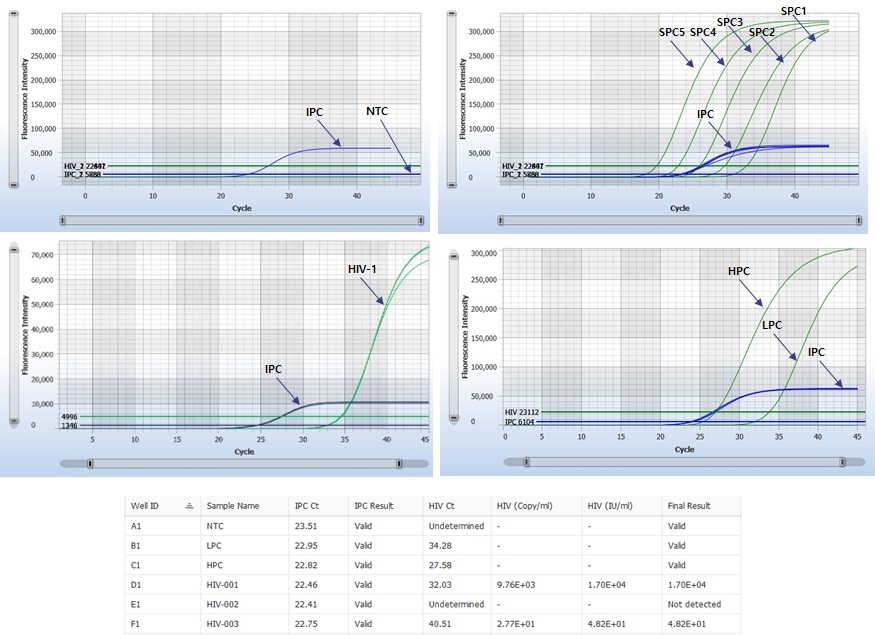
AccuPower® HIV-1 Quant Kit (ExiStation™ FA 96) enables quantitative detection of HIV-1 RNA from 50 IU/mL to 10⁸ IU/mL.
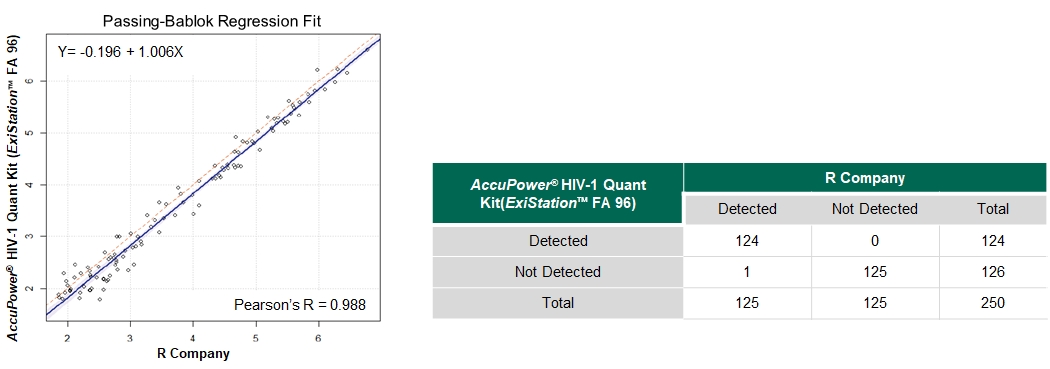
- Clinical Sensitivity = 99.2% (95% CI: 95.6 to 99.9)
- Clinical Specificity = 100.0% (95% CI: 97.0 to 100.0)
The clinical evaluation of the AccuPower® HIV-1 Quant Kit (ExiStation™ FA 96) was conducted using 250 residual clinical specimens collected in Europe.
Ordering Information
| Cat. No. | Product Description | Certification |
| HIV-3116 | AccuPower® HIV-1 Quant Kit (ExiStation™ FA 96) | CE-IVDR Class D |
Related Products
| Cat. No. | Instrument |
| A-2500-1 | ExiStation™ FA 96 |
| Cat. No. | Nucleic Acid Extraction Kit |
| K-4814 | ExiStation™ FA DNA/RNA Kit(V2) |
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
Contact Us
E-mail : sales@bioneer.com
v0.0 / 2019-03-05