We use cookies to give you the best online experience. By using our website you agree to our use of cookies in accordance with our Cookie Policy
AccuPower® CMV Quantitative PCR Kit
Human cytomegalovirus (HCMV) infection is acquired subclinically during childhood. It is usually transmitted via body fluid, blood, solid organ transplant and stem cell transplant recipients. It causes fever, splenomegaly, liver dysfunction or atypical lymph node hyperplasia. Thus, monitoring of CMV viral load is essential for newborns and transplant recipients.
Features and Benefits
- Simplified Procedure: Whole blood, serum, plasma and breast milk samples can be directly used for the quantification of CMV viral load using ExiStation™ MDx System.
- Signature Convenience: All components for the assay are contained within a tube.
- Enhanced Sensitivity & Specificity: Bioneer’s proprietary HotStart technology accomplishes high sensitivity and specificity.
- Remarkable Stability: Vacuum-dried premix allows stable and reproducible results.
- High Quality: All diagnostic kits manufactured by Bioneer are under strict quality control guidelines.
Specifications
| Specimen Type | Whole blood, EDTA-plasma, serum, breast milk |
| Kit Contents | PCR Premix, SPC 1~5, IPC, NTC, SL buffer, DEPC D.W. |
| Instrumentation | ExiStation™ Universal Molecular Diagnostic System or Exicycler™ 96 |
| Tests | 96 |
Performances
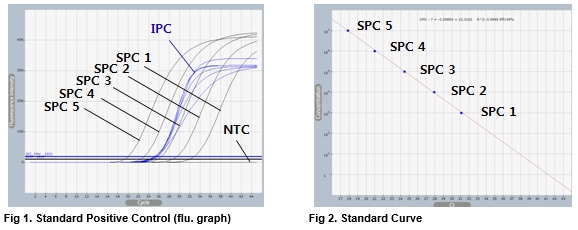
AccuPower® CMV Quantitative PCR Kit includes serially diluted Standard Positive Control (SPC) 1~5 for the quantification of CMV.
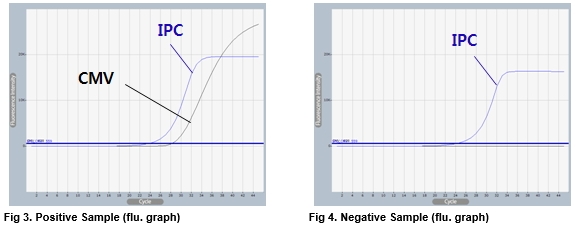
AccuPower® CMV Quantitative PCR Kit test results using clinical samples. The kit employs IPC in all wells to confirm correct PCR amplification.
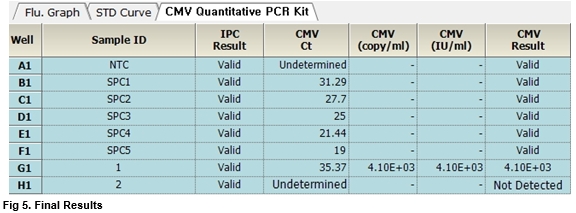
User centric ExiStation Manager software or ExiDiagnosis software automatically analyzes the test results based upon the Ct (threshold cycle) value.
Ordering Information
| Cat. No. | Product Description |
| CMV-1111 | AccuPower® CMV Quantitative PCR Kit (96 tests) |
Related Products
| Cat. No. | MDx Diagnostic Kit |
| BKV-1111 | AccuPower® BKV Quantitative PCR Kit (96 tests) |
| EBV-1111 | AccuPower® EBV Quantitative PCR Kit (96 tests) |
| Cat. No. | Instrument |
| A-2060 | Exicycler™ 96 Real-Time Quantitative Thermal Block |
| A-2200 | ExiStation™ Universal Molecular Diagnostic System |
| A-5050 | ExiPrep™16 Dx Automated Nucleic Acid Extraction System (16 tests) |
| Cat. No. | Nucleic Acid Extraction Kit |
| K-4471 | ExiPrep™ Dx Viral DNA/RNA Kit (96 tests) |
| K-4472 | ExiPrep™ Dx Viral DNA Kit (96 tests) |
| K-4474 | ExiPrep™ Dx Blood Viral DNA Kit (96 test) |
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
Contact Us
E-mail : sales@bioneer.com