We use cookies to give you the best online experience. By using our website you agree to our use of cookies in accordance with our Cookie Policy
ExiPrep™ Dx Viral RNA Kit
Features and Benefits
1. Simultaneous extraction of nucleic acids from up to 16 clinical samples
2. Optimized protocols for nucleic acid extraction according to sample types are pre-installed for reproducible results.
3. ExiPrep™16 Dx system uses silica magnetic beads developed and produced with Bioneer’s technology.
4. Pre-filled buffer cartridge system
Specifications
| Technology | Magnetic Silica Beads |
| Main sample type | Plasma, serum, swab, urine, stool, CSF |
| Starting volume | 400 ㎕ |
| Elution volume | 50 ㎕ |
| Instrumentation | ExiPrep™16 Dx Fully Automated Nucleic Acid Extraction System |
| Tests | 96 |
| Certification | CE, MFDS |
Applications
Diagnosis of infectious disease based on real-time PCR:
HCV, Norovirus, Enterovirus, Influenza virus, etc.
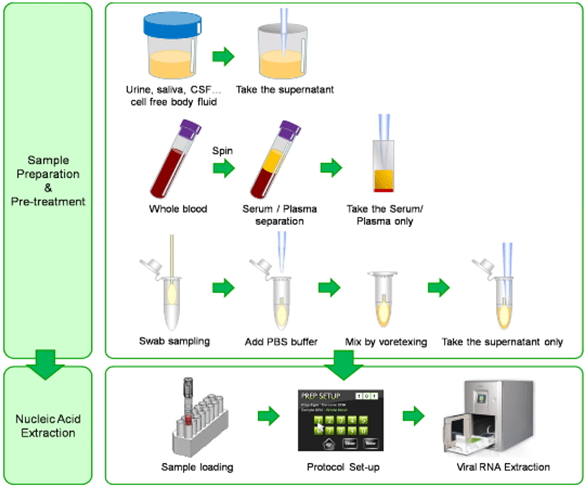
Workflow
Performances
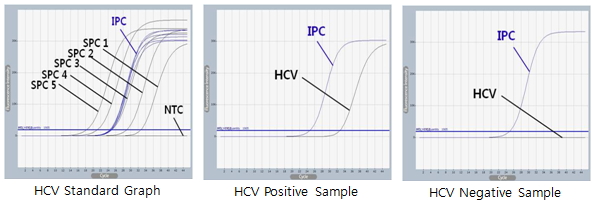
Figure 1. Quantification of HCV viral load was performed to evaluate ExiPrep™ Dx Viral RNA Kit. Viral RNA was extracted from clinical serum using the kit. Real-time PCR was performed with the AccuPower® HCV Quantitative RT-PCR Kit on Exicycler™ 96.
Ordering Information
| Cat. No. | Product Description |
| K-4473 | ExiPrep™ Dx Viral RNA Kit (96 tests) |
Related Products
| Cat. No. | Instrument |
| A-5050 | ExiPrep™16 Dx Automated Nucleic Acid Extraction System (16 tests) |
| Cat. No. | MDx Kits |
| E71-1111 | AccuPower® Enterovirus 71 Real-Time RT-PCR Kit (96 tests) |
| ENT-1111 | AccuPower® EV Real-Time RT-PCR Kit (96 tests) |
| HCV-1111 | AccuPower® HCV Quantitative RT-PCR Kit (96 tests) |
| IFA-1111 | AccuPower® Influenza A Real-Time RT-PCR Kit (96 tests) |
| NOR-1111 | AccuPower® Norovirus Real-Time RT-PCR Kit (96 tests) |
| SIA-1111 | AccuPower® new Inf A (H1N1) & Inf A Real-Time RT-PCR Kit (48 tests) |
| SIV-1111 | AccuPower® new Inf A (H1N1) Real-Time RT-PCR Kit (96 tests) |
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
Contact Us
E-mail : sales@bioneer.com